Unlock Physics 5054 Solutions: Past Papers & Guidelines for A
Physics is not just a subject—it’s a gateway to understanding the universe and unlocking endless possibilities for your future. For students pursuing Physics 5054, the journey to mastery can feel challenging, but it’s also immensely rewarding. Whether you’re striving to solve complex problems, grasp intricate theories, or perfect your practical skills, having access to the right resources can make all the difference.
If you’ve been searching for Physics 5054 past papers, lesson plans, activities, tutors, or any other resource, you’ve come to the right place. Let’s embark on this transformative journey and uncover the secrets to scoring an A* in Physics 5054.
Why Physics 5054 Is More Than Just an Exam
Physics 5054 is a Cambridge O Level subject that bridges the gap between theoretical understanding and real-world application. It introduces students to the principles that govern energy, matter, and the physical world, laying the foundation for future careers in engineering, technology, research, and beyond.
The Key Challenges in Physics 5054
- Complex Theories: Concepts like electromagnetism, thermodynamics, and waves can be difficult to master without the right support.
- Time Management: Balancing syllabus coverage, revision, and practical skills is no small feat.
- Lack of Quality Resources: Many students struggle to find reliable Physics 5054 past papers, solved papers, and up-to-date notes.
Fortunately, platforms like CambridgeClassroom.com are revolutionizing how students and teachers approach Physics 5054 by providing all the tools you need in one place.
The Secret to Scoring A in Physics 5054*
1. Master the Basics with Physics 5054 Notes
Strong fundamentals are the cornerstone of success. Use Physics 5054 notes to:
- Understand core concepts in a simplified manner.
- Break down complex theories into manageable chunks.
- Create a strong foundation for tackling advanced problems.
Where to Find Help: CambridgeClassroom.com offers comprehensive, easy-to-understand notes tailored to the Physics 5054 latest syllabus.
2. Practice with Physics 5054 Past Papers and Solved Papers
Past papers are your best friend when it comes to exam preparation. They help you:
- Familiarize yourself with the exam format.
- Identify recurring topics and question patterns.
- Practice time management under exam conditions.
To take your preparation further, Physics 5054 solved papers provide:
- Step-by-step solutions to challenging problems.
- Insights into examiner expectations.
- Strategies to avoid common mistakes.
3. Organize Your Learning with Physics 5054 Lesson Plans
Structured learning is the key to consistent progress. Physics 5054 lesson plans help students and tutors:
- Cover the syllabus systematically.
- Allocate time effectively to each topic.
- Ensure no concept is left behind.
Educators will find pre-designed lesson plans on CambridgeClassroom.com that are tailored for the Physics 5054 latest syllabus, saving time and enhancing teaching quality.
4. Engage with Physics 5054 Activities
Physics is best understood through hands-on experiences. Interactive Physics 5054 activities bring concepts to life, making learning:
- Engaging and memorable.
- Relevant to real-world scenarios.
- Practical, especially for experimental questions.
Explore CambridgeClassroom.com for a variety of activities and simulations designed to align with the Physics 5054 guidelines.
5. Seek Expert Guidance with Physics 5054 Tutors and Tuitions
Sometimes, a little extra help is all you need to unlock your full potential. Expert Physics 5054 tutors can:
- Provide personalized attention to address your weaknesses.
- Offer tailored advice and strategies to boost your performance.
- Keep you motivated and focused on your goals.
For flexible learning, many students opt for Physics 5054 online help or tuitions. CambridgeClassroom.com connects students with experienced tutors who specialize in guiding learners toward achieving an A*.
6. Test Yourself with Physics 5054 Worksheets
Physics 5054 worksheets are excellent tools for self-assessment and reinforcement. They allow students to:
- Practice a variety of question types.
- Hone their problem-solving skills.
- Identify areas that need improvement.
Worksheets on CambridgeClassroom.com are carefully designed to challenge students while reinforcing their knowledge.
7. Follow the Physics 5054 Guidelines and Syllabus Closely
The Physics 5054 latest syllabus is your roadmap to success. Make sure you:
- Review the syllabus frequently to ensure all topics are covered.
- Align your preparation with the guidelines provided by Cambridge.
- Use resources like marking schemes to understand how answers should be structured.
How CambridgeClassroom.com Can Transform Your Physics 5054 Preparation
If you’ve been searching for the ultimate resource hub for Physics 5054 past papers, lesson plans, activities, and more, look no further than CambridgeClassroom.com. Here’s why it’s the best choice for students and educators:
1. Comprehensive Resource Library
CambridgeClassroom.com offers:
- An extensive collection of Physics 5054 past papers and marking schemes.
- Solved papers with detailed solutions.
- Worksheets, notes, and lesson plans tailored to the syllabus.
2. Easy-to-Navigate Platform
With a user-friendly interface, you can:
- Quickly search for the resources you need.
- Download materials in PDF or ZIP formats.
- Access resources anytime, anywhere.
3. Regular Updates
All resources are regularly updated to align with the Physics 5054 latest syllabus, ensuring you’re always working with the most relevant materials.
4. Affordable and Accessible
Many essential resources are free, with affordable premium options for advanced tools.
5. Expert Support
From connecting with top Physics 5054 tutors to accessing personalized online help, CambridgeClassroom.com ensures you have all the support you need to excel.
Tips to Make the Most of Your Physics 5054 Preparation
- Start Early: Don’t wait until the last minute. Begin your preparation well in advance to avoid unnecessary stress.
- Set Goals: Break the syllabus into manageable sections and set achievable goals for each study session.
- Stay Consistent: Regular practice with Physics 5054 worksheets, activities, and past papers will reinforce your learning.
- Analyze Your Performance: Review your mistakes and focus on areas that need improvement.
- Seek Guidance: Don’t hesitate to reach out to tutors or use online help if you’re struggling with specific topics.
Success Stories: Students Who Scored A in Physics 5054*
“I never thought I’d enjoy Physics 5054, but CambridgeClassroom.com made it so much easier. The solved papers were a lifesaver!” – Maria, O Level Student
_“The lesson plans helped me stay on track, and the activities made learning fun. I scored an A!”_* – Ahmed, O Level Candidate
“As a teacher, I rely on CambridgeClassroom.com for everything—from worksheets to lesson plans. It’s an invaluable resource.” – Mr. Tan, Physics Educator
FAQs About Physics 5054 Preparation
1. Where can I find Physics 5054 past papers and solved papers?
You can access a comprehensive collection of past papers and solved papers on CambridgeClassroom.com.
2. What are the best tools for Physics 5054 self-study?
Use Physics 5054 notes, worksheets, and marking schemes to reinforce your understanding and practice problem-solving.
3. How can I make Physics 5054 lessons more engaging?
Incorporate interactive activities and experiments to bring concepts to life. CambridgeClassroom.com offers a variety of resources for this purpose.
4. Are there online tutors for Physics 5054?
Yes, CambridgeClassroom.com connects students with experienced Physics 5054 tutors for personalized guidance.
5. How do I stay updated with the Physics 5054 latest syllabus?
Regularly review the syllabus and use CambridgeClassroom.com for updated materials aligned with the latest guidelines.
Conclusion: Your A Awaits*
Success in Physics 5054 begins with the right preparation, and CambridgeClassroom.com has everything you need to excel. From past papers to lesson plans, activities, and tutors, it’s your one-stop solution for academic excellence.
Start your journey today and take the first step toward achieving an A* in Physics 5054. Visit www.cambridgeclassroom.com now!




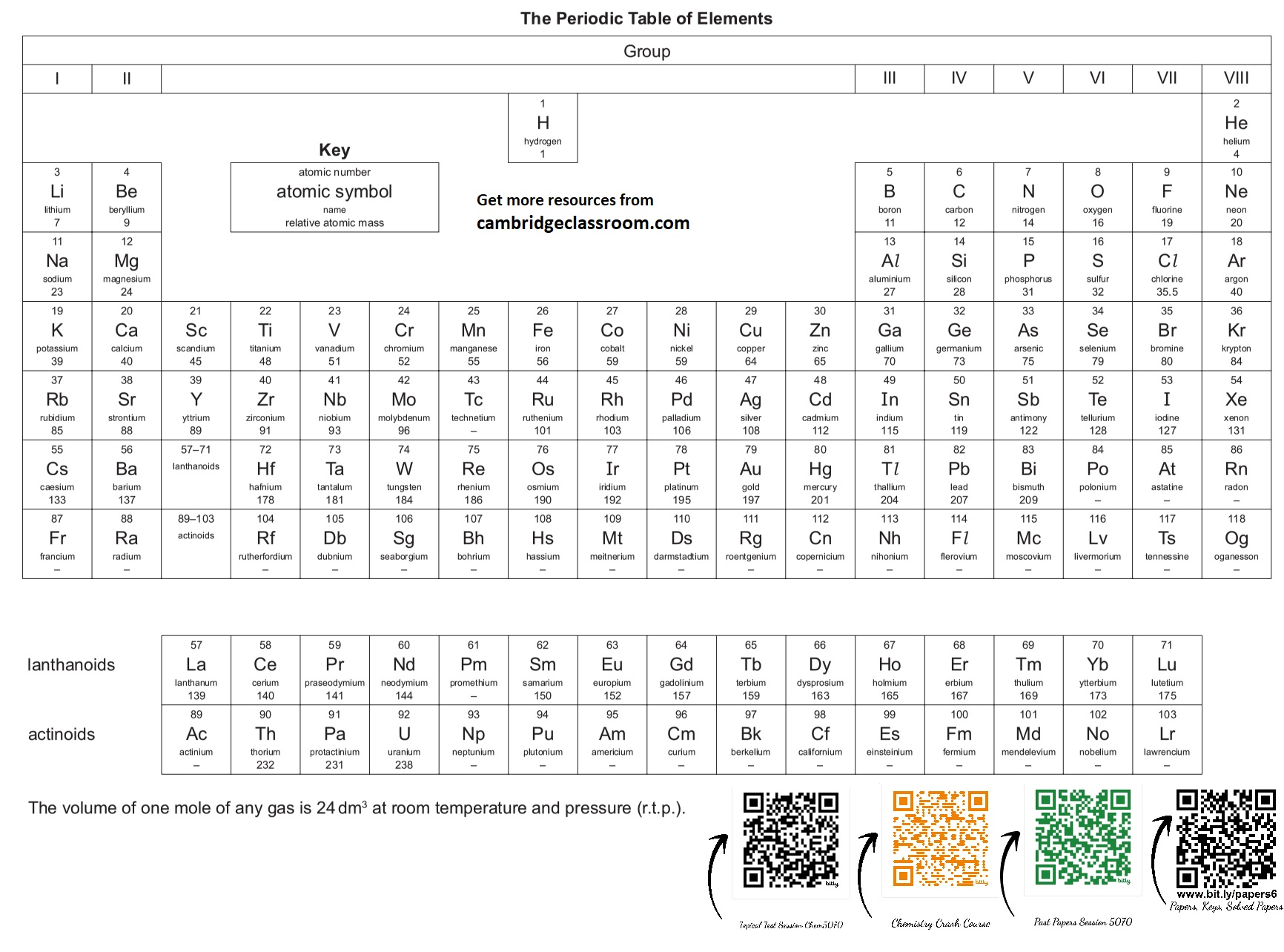





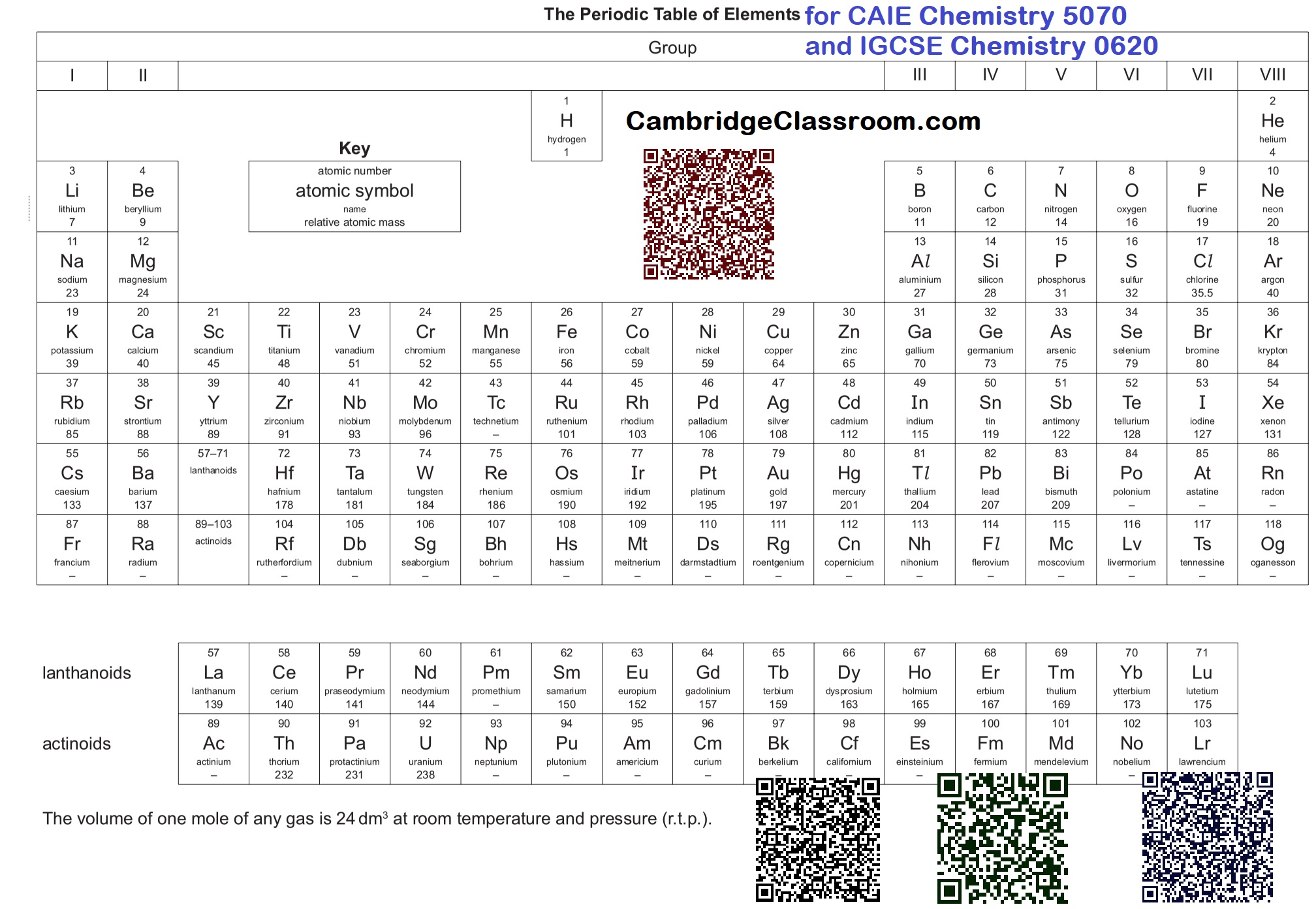









![Fundamental Concepts & States of Matter • Atom: The smallest particle of an element that can exist, made of a nucleus (protons and neutrons) and electrons orbiting it. • Element: A pure substance consisting of only one type of atom, which cannot be broken down into simpler substances by chemical means. • Compound: A substance formed when two or more different elements are chemically bonded together in a fixed ratio. • Mixture: A substance containing two or more elements or compounds not chemically bonded together. Can be separated by physical means. • Molecule: A group of two or more atoms held together by chemical bonds. • Proton: A subatomic particle found in the nucleus with a relative mass of 1 and a charge of +1. • Neutron: A subatomic particle found in the nucleus with a relative mass of 1 and no charge (0). • Electron: A subatomic particle orbiting the nucleus with a negligible relative mass and a charge of -1. • Atomic Number (Z): The number of protons in the nucleus of an atom. Defines the element. • Mass Number (A): The total number of protons and neutrons in the nucleus of an atom. • Isotopes: Atoms of the same element (same atomic number) but with different mass numbers due to a different number of neutrons. • Relative Atomic Mass ($A_r$): The weighted average mass of an atom of an element compared to $1/12$th the mass of a carbon-12 atom. • Relative Molecular Mass ($M_r$): The sum of the relative atomic masses of all atoms in one molecule of a compound. • Relative Formula Mass ($M_r$): The sum of the relative atomic masses of all atoms in the formula unit of an ionic compound. • Mole: The amount of substance that contains $6.02 \times 10^{23}$ particles (Avogadro's number). • Molar Mass: The mass of one mole of a substance, expressed in g/mol. Numerically equal to $A_r$ or $M_r$. • Empirical Formula: The simplest whole number ratio of atoms of each element in a compound. • Molecular Formula: The actual number of atoms of each element in a molecule. • Solid: Particles are closely packed in a fixed, regular arrangement, vibrate about fixed positions. Definite shape and volume. • Liquid: Particles are closely packed but randomly arranged, can slide past each other. Definite volume, no definite shape. • Gas: Particles are far apart and arranged randomly, move rapidly and randomly. No definite shape or volume. • Melting Point: The specific temperature at which a solid changes into a liquid at a given pressure. • Boiling Point: The specific temperature at which a liquid changes into a gas (vaporizes) at a given pressure. • Sublimation: The direct change of state from solid to gas without passing through the liquid phase (e.g., solid $\text{CO}_2$). • Diffusion: The net movement of particles from a region of higher concentration to a region of lower concentration, due to random motion. • Osmosis: The net movement of water molecules across a partially permeable membrane from a region of higher water potential to a region of lower water potential. 2. Structure & Bonding • Ionic Bond: The electrostatic force of attraction between oppositely charged ions, formed by the transfer of electrons from a metal to a non-metal. • Covalent Bond: A strong electrostatic force of attraction between a shared pair of electrons and the nuclei of the bonded atoms, typically between two non-metals. • Metallic Bond: The electrostatic force of attraction between positive metal ions and delocalised electrons. • Ion: An atom or group of atoms that has gained or lost electrons, resulting in a net electrical charge. • Cation: A positively charged ion (lost electrons). • Anion: A negatively charged ion (gained electrons). • Octet Rule: Atoms tend to gain, lose, or share electrons in order to achieve a full outer electron shell, typically with eight electrons. • Giant Ionic Lattice: A regular, repeating 3D arrangement of oppositely charged ions, held together by strong electrostatic forces. • Simple Molecular Structure: Molecules held together by strong covalent bonds, but with weak intermolecular forces between molecules. • Giant Covalent Structure (Macromolecular): A large structure where all atoms are held together by strong covalent bonds in a continuous network (e.g., diamond, silicon dioxide). • Allotropes: Different structural forms of the same element in the same physical state (e.g., diamond and graphite are allotropes of carbon). • Electronegativity: The power of an atom to attract the electron pair in a covalent bond to itself. • Polar Covalent Bond: A covalent bond in which electrons are shared unequally due to a difference in electronegativity between the bonded atoms. • Hydrogen Bond: A strong type of intermolecular force that occurs between molecules containing hydrogen bonded to a highly electronegative atom (N, O, F). • Van der Waals' forces: Weak intermolecular forces of attraction between all molecules, arising from temporary dipoles. 3. Stoichiometry & Chemical Calculations • Stoichiometry: The study of quantitative relationships between reactants and products in chemical reactions. • Limiting Reactant: The reactant that is completely consumed in a chemical reaction, determining the maximum amount of product that can be formed. • Excess Reactant: The reactant present in a greater amount than required to react with the limiting reactant. • Yield: The amount of product obtained from a chemical reaction. • Theoretical Yield: The maximum amount of product that can be formed from a given amount of reactants, calculated using stoichiometry. • Actual Yield: The amount of product actually obtained from a chemical reaction, usually less than the theoretical yield. • Percentage Yield: $($Actual Yield $/$ Theoretical Yield$) \times 100\%$. • Concentration: The amount of solute dissolved in a given volume of solvent or solution. Often expressed in mol/dm$^3$ (molarity) or g/dm$^3$. • Solute: The substance that dissolves in a solvent to form a solution. • Solvent: The substance in which a solute dissolves to form a solution. • Solution: A homogeneous mixture formed when a solute dissolves in a solvent. 4. Chemical Reactions & Energetics • Chemical Reaction: A process that involves rearrangement of the atomic structure of substances, resulting in the formation of new substances. • Reactants: The starting substances in a chemical reaction. • Products: The substances formed as a result of a chemical reaction. • Word Equation: An equation that uses the names of the reactants and products. • Symbol Equation: An equation that uses chemical symbols and formulae to represent reactants and products, and is balanced. • Balancing Equation: Ensuring the number of atoms of each element is the same on both sides of a chemical equation. • Redox Reaction: A reaction involving both reduction and oxidation. • Oxidation: Loss of electrons, gain of oxygen, or loss of hydrogen. Increase in oxidation state. • Reduction: Gain of electrons, loss of oxygen, or gain of hydrogen. Decrease in oxidation state. • Oxidising Agent: A substance that causes oxidation by accepting electrons (and is itself reduced). • Reducing Agent: A substance that causes reduction by donating electrons (and is itself oxidised). • Exothermic Reaction: A reaction that releases energy to the surroundings, usually as heat, causing the temperature of the surroundings to rise. $\Delta H$ is negative. • Endothermic Reaction: A reaction that absorbs energy from the surroundings, usually as heat, causing the temperature of the surroundings to fall. $\Delta H$ is positive. • Activation Energy ($E_a$): The minimum amount of energy required for reactants to collide effectively and initiate a chemical reaction. • Catalyst: A substance that increases the rate of a chemical reaction without being chemically changed itself, by providing an alternative reaction pathway with a lower activation energy. • Enthalpy Change ($\Delta H$): The heat energy change measured at constant pressure for a reaction. • Standard Enthalpy of Formation ($\Delta H_f^\circ$): The enthalpy change when one mole of a compound is formed from its constituent elements in their standard states under standard conditions. • Standard Enthalpy of Combustion ($\Delta H_c^\circ$): The enthalpy change when one mole of a substance is completely combusted in oxygen under standard conditions. • Hess's Law: The total enthalpy change for a reaction is independent of the route taken, provided the initial and final conditions are the same. 5. Rates of Reaction & Equilibrium • Rate of Reaction: The change in concentration of a reactant or product per unit time. • Collision Theory: For a reaction to occur, reactant particles must collide with sufficient energy (activation energy) and correct orientation. • Factors Affecting Rate: Concentration, pressure (for gases), surface area, temperature, and presence of a catalyst. • Reversible Reaction: A reaction where products can react to reform the original reactants, indicated by $\rightleftharpoons$. • Chemical Equilibrium: A state in a reversible reaction where the rate of the forward reaction is equal to the rate of the reverse reaction, and the concentrations of reactants and products remain constant. • Le Chatelier's Principle: If a change in conditions (temperature, pressure, concentration) is applied to a system at equilibrium, the system will shift in a direction that counteracts the change. 6. Acids, Bases & Salts • Acid: A substance that produces hydrogen ions ($H^+$) when dissolved in water (Arrhenius definition) or a proton donor (Brønsted-Lowry definition). • Base: A substance that produces hydroxide ions ($OH^-$) when dissolved in water (Arrhenius definition) or a proton acceptor (Brønsted-Lowry definition). • Alkali: A soluble base that dissolves in water to produce hydroxide ions ($OH^-$). • Salt: A compound formed when the hydrogen ion of an acid is replaced by a metal ion or an ammonium ion. • Neutralisation: The reaction between an acid and a base (or alkali) to form a salt and water. $H^+(aq) + OH^-(aq) \rightarrow H_2O(l)$. • pH: A measure of the acidity or alkalinity of a solution, defined as $-\log_{10}[H^+]$. Scale from 0 to 14. • Strong Acid: An acid that fully dissociates (ionizes) in water (e.g., HCl, $H_2SO_4$). • Weak Acid: An acid that partially dissociates (ionizes) in water (e.g., $CH_3COOH$). • Strong Base: A base that fully dissociates in water (e.g., NaOH, KOH). • Weak Base: A base that partially dissociates in water (e.g., $NH_3$). • Amphoteric: A substance that can act as both an acid and a base (e.g., aluminium oxide, water). • Titration: A quantitative chemical analysis method used to determine the unknown concentration of a reactant using a known concentration of another reactant. • Indicator: A substance that changes colour over a specific pH range, used to detect the endpoint of a titration. 7. Electrochemistry • Electrolysis: The decomposition of an ionic compound using electrical energy. Requires molten or aqueous electrolyte. • Electrolyte: An ionic compound (molten or dissolved in a solvent) that conducts electricity due to the movement of ions. • Electrodes: Conductors (usually metal or graphite) through which electricity enters and leaves the electrolyte. • Anode: The positive electrode, where oxidation occurs (anions are attracted). • Cathode: The negative electrode, where reduction occurs (cations are attracted). • Faraday's Laws of Electrolysis: Relate the amount of substance produced at an electrode to the quantity of electricity passed through the electrolyte. • Galvanic (Voltaic) Cell: An electrochemical cell that generates electrical energy from spontaneous redox reactions. • Standard Electrode Potential ($E^\circ$): The potential difference of a half-cell compared to a standard hydrogen electrode under standard conditions (1 M concentration, 1 atm pressure for gases, 298 K). • Electrochemical Series: A list of elements arranged in order of their standard electrode potentials, indicating their relative reactivity as oxidising or reducing agents. 8. The Periodic Table • Periodic Table: An arrangement of elements in order of increasing atomic number, showing periodic trends in properties. • Group: A vertical column in the periodic table, containing elements with the same number of valence electrons and similar chemical properties. • Period: A horizontal row in the periodic table, containing elements with the same number of electron shells. • Valence Electrons: Electrons in the outermost shell of an atom, involved in chemical bonding. • Alkali Metals (Group 1): Highly reactive metals, readily lose one electron to form $+1$ ions. React vigorously with water. • Alkaline Earth Metals (Group 2): Reactive metals, readily lose two electrons to form $+2$ ions. • Halogens (Group 17/7): Highly reactive non-metals, readily gain one electron to form $-1$ ions. Exist as diatomic molecules. • Noble Gases (Group 18/0): Unreactive elements with a full outer electron shell, existing as monatomic gases. • Transition Metals: Elements in the d-block of the periodic table, characterised by variable oxidation states, coloured compounds, and catalytic activity. • Metallic Character: Tendency of an element to lose electrons and form positive ions. Increases down a group, decreases across a period. • Non-metallic Character: Tendency of an element to gain electrons and form negative ions. Decreases down a group, increases across a period. • Ionisation Energy: The energy required to remove one electron from each atom in one mole of gaseous atoms to form one mole of gaseous $1+$ ions. • Electron Affinity: The energy change when one mole of electrons is added to one mole of gaseous atoms to form one mole of gaseous $1-$ ions. 9. Organic Chemistry • Organic Chemistry: The study of carbon compounds, excluding carbonates, carbides, and oxides of carbon. • Hydrocarbon: A compound containing only carbon and hydrogen atoms. • Saturated Hydrocarbon: A hydrocarbon containing only single carbon-carbon bonds (e.g., alkanes). • Unsaturated Hydrocarbon: A hydrocarbon containing one or more carbon-carbon double or triple bonds (e.g., alkenes, alkynes). • Homologous Series: A series of organic compounds with the same general formula, similar chemical properties, and showing a gradual change in physical properties. • Functional Group: A specific group of atoms within a molecule that is responsible for the characteristic chemical reactions of that molecule. • Alkane: Saturated hydrocarbons with the general formula $C_nH_{2n+2}$. Contain only single bonds. • Alkene: Unsaturated hydrocarbons with the general formula $C_nH_{2n}$. Contain at least one carbon-carbon double bond. • Alkyne: Unsaturated hydrocarbons with the general formula $C_nH_{2n-2}$. Contain at least one carbon-carbon triple bond. • Alcohol: Organic compounds containing the hydroxyl functional group ($-OH$). General formula $C_nH_{2n+1}OH$. • Carboxylic Acid: Organic compounds containing the carboxyl functional group ($-COOH$). • Ester: Organic compounds formed from the reaction of a carboxylic acid and an alcohol, containing the ester linkage ($-COO-$). • Isomers: Compounds with the same molecular formula but different structural formulae. • Structural Isomers: Isomers that differ in the arrangement of their atoms or bonds. • Addition Reaction: A reaction where an unsaturated molecule adds across a double or triple bond, forming a single product. • Substitution Reaction: A reaction where an atom or group of atoms in a molecule is replaced by another atom or group of atoms. • Polymerisation: The process of joining many small monomer molecules together to form a large polymer molecule. • Monomer: A small molecule that can be joined together to form a polymer. • Polymer: A large molecule (macromolecule) formed from many repeating monomer units. • Addition Polymerisation: Polymerisation where monomers add to one another in such a way that the polymer contains all the atoms of the monomer. Usually involves unsaturated monomers. • Condensation Polymerisation: Polymerisation where monomers join together with the elimination of a small molecule (e.g., water). • Cracking: The process of breaking down long-chain hydrocarbons into shorter, more useful hydrocarbons using heat and/or a catalyst. • Fermentation: The anaerobic respiration of yeast, converting glucose into ethanol and carbon dioxide. 10. Analytical Chemistry • Qualitative Analysis: The identification of the components of a sample. • Quantitative Analysis: The determination of the amount or concentration of a component in a sample. • Chromatography: A separation technique based on differential partitioning between a stationary phase and a mobile phase. • Retention Factor ($R_f$): In paper/thin-layer chromatography, the ratio of the distance travelled by the spot to the distance travelled by the solvent front. • Spectroscopy: The study of the interaction of electromagnetic radiation with matter. • Infrared (IR) Spectroscopy: Used to identify functional groups in organic molecules based on their absorption of IR radiation. • Mass Spectrometry: Used to determine the relative molecular mass of a compound and its fragmentation pattern to deduce structure. • Flame Test: A qualitative test for the presence of certain metal ions, which produce characteristic colours when heated in a flame.](https://cambridgeclassroom.com/wp-content/uploads/2024/03/White-And-Purple-Modern-Online-Graphic-Design-Courses-Instagram-Post-4.png)




